The therascreen EGFR test covers more clinically validated mutations to deliver the information you need to make the best therapeutic decisions.
Focus on what matters
The therascreen EGFR RGQ PCR Kit is the only FDA-approved and clinically validated assay for the selection of patients with NSCLC for whom GILOTRIF or IRESSA are indicated.
Clinically actionable information is what helps treat patients
Detect more clinically validated (15) and analytically validated (21) mutations than with any other commercial assay
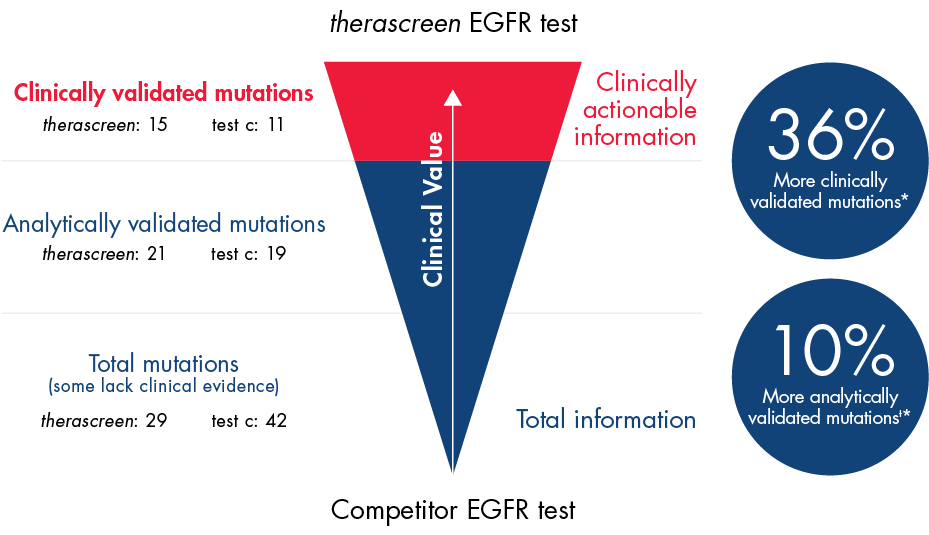
* Analytically validated mutations with clinical outcome data from LUX-Lung 3, IFUM, AURA2 and EURTAC clinical trials.
†S therascreen EGFR RGQ PCR Kit Instructions for Use (Handbook)SDownload handbook
Contact us now!
QIAGEN’s lung cancer solutions offer many benefits, including convenient, efficient and precise EGFR testing, reliable, rapid and cost effective workflows, and secure systems with experienced services and support.
Lung cancer EGFR — Oncologists
The therascreen EGFR RGQ PCR Kit (therascreen EGFR test) reliably detects mutations in the EGFR oncogene which are found in lung cancer. EGFR testing is used to select NSCLC patients who are being considered for treatment with either GILOTRIF (afatinib) or IRESSA (gefitinib).
Lab finder — therascreen EGFR test
QIAGEN’s therascreen mutation detection tests help determine what treatment options may be best for cancer patients. Find out which labs are using the therascreen EGFR RGQ PCR Kit and other therascreen biomarker tests!
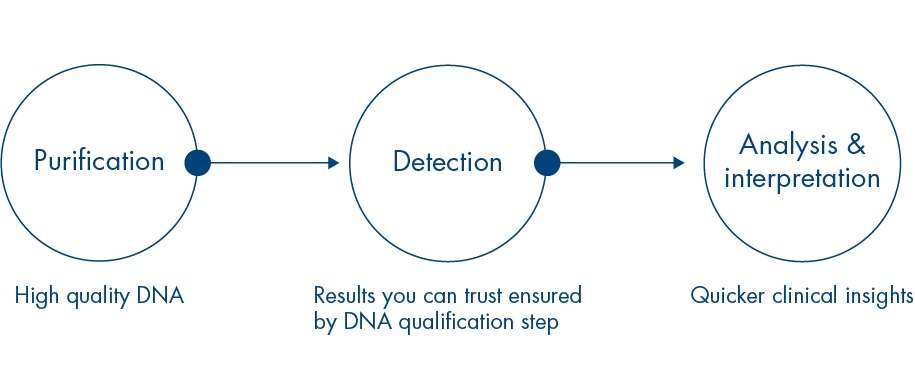
The therascreen EGFR Sample to Insight workflow provides quick and reliable results you and your patients can have confidence in.